Microdosing TRT – The Future of Testosterone Replacement Therapy

Introduction
The premise behind microdosing Testosterone Replacement Therapy (TRT) is quite simple; it is using the minimum effective dose, in the most effective manner, in order to achieve the desired outcome which, in this case, is stable male androgen levels.
Our gold standard TRT is daily subcutaneous Testosterone Cypionate (or Enanthate) and Human Chorionic Gonadotropin (HCG) injections. Although we are a private clinic, and therefore perhaps not as constrained in our choice of treatment as the NHS, we are delighted to have shared care arrangements with a rapidly growing number of GPs and NHS Consultants, who have agreed to prescribe their patient’s Testosterone medication and support their treatment through the NHS, appreciating the importance of their patient’s day to day well-being and long term health.
Whilst Testosterone Cypionate is more suitable for subcutaneous injections and clinically our first choice of Testosterone ester (something discussed further in this article), from a half-life perspective, Cypionate and Enanthate are interchangeable. This does not mean that it’s impossible to achieve stable levels with other Testosterone esters however. On the contrary, we have also experienced positive results with patients using Testosterone Undecanoate, the difference being that we use a more frequent microdose to achieve stability.
Current prescribing recommendations for the testosterone preparations available are disappointingly out-dated, with the NHS adopting a one-size-fits-all approach to TRT in most instances. By choosing to prescribe outside of the manufacturers guidelines (off licence) and instead microdosing Testosterone, we see less side effects because we are avoiding the peaks and troughs that are often inevitable with other treatment regimes. Unsurprisingly, your body has no other option than to react accordingly to the current level of Testosterone in your body. Through application of basic pharmacokinetic principles, alongside the currently available research, we aim to demonstrate why we have found microdosing to be the most effective way of achieving hormonal balance, without fluctuations.
Despite there being British, European and International guidelines to help support clinicians in diagnosing and managing Testosterone Deficiency (TD), there still appears to be a strong resistance to change within the medical community. In the past, we have been vehemently challenged by NHS Endocrinologists, specifically one who is also a Media Ambassador for the Endocrinology Society and GMC Expert, something that I discussed further in ‘TRT – Best Practice’. The reasons for such resistance are multi-factorial, however we are determined to continue to affect a positive change to how Testosterone Deficiency (TD) is perceived and treated within the UK healthcare system. There must be a reason why men travel to our clinic from all over the UK, Europe and other locations across the globe. We believe that transparency is integral to trust, and our outcomes speak for themselves.
Classically, men present to our clinic with a multitude of symptoms. There is typically an element of mental discord (ranging from minor to severe), troublesome lethargy, a decline in cognitive function often described as ‘brain fog’, and low libido. The quantitative signs such as erectile dysfunction, loss of lean muscle mass and increased adiposity are also important, but most men are simply seeking a semblance of normality.
It must be noted that we never advocate high, abnormal Testosterone levels. The primary purpose of Testosterone is arguably to help facilitate growth and repair; to recover from the day and prepare for tomorrow. It is important to appreciate that all hormones are dependent, whether that be a direct or indirect relationship, they are not independent. An excess of any one hormone has a cascade effect on other physiological parameters. I have often lowered a patients self-perceived “optimal” levels, for them to report that their anxiety has settled and that their libidos have improved.
It is also important to understand natural Testosterone production within the body and the role of Testosterone and its metabolites, namely Dihydrotestosterone (DHT) and Oestradiol (E2), in long term physical and psychological well-being. To affect a sustainable change, we must first attempt to mimic natural physiology with TRT, with an appreciation that Testosterone merely serves as a foundation to help facilitate bodily functions. Not only do we need to adopt a personalised approach to patient-centred care, we need to appreciate the need for a holistic approach to medicine. TRT will not reverse the negative symptoms that the patient is experiencing if we do not support them in other aspects of their life. Hormones are not aware of what day of the week it is, or how far away you are from your next Testosterone dose, they follow a strict 24-hour circadian pattern, which is discussed in further detail in ‘The Endocrine System & Sleep’.
We will now give you a brief background into pharmacokinetics, prior to demonstrating what the everyday environment can look like on different treatment protocols. Be under no illusion that this is a guide on “How To Prescribe TRT”, there are numerous complexities to achieving and sustaining hormonal balance that must be taken into consideration and regularly reviewed. These include physiological parameters, body composition, personal requirements and so on. Your TRT protocol must ALWAYS be under the supervision and guidance of a doctor specialising in TRT.
Pharmacokinetics
Pharmacokinetics, the name arises from ancient Greek; pharmakon “drug” and kinetikos “moving or putting in motion”. It is the study of how an organism affects a drug. In the case of you and I, we can describe it as the movement of a drug we’ve been prescribed, into, through, and out of our body, and the time course of its absorption, bioavailability, distribution, metabolism and excretion. We perform these steps all at a different rate and manner due to our different genetic profiles and environmental differences. These differences can help explain why you may know of a friend who can drink nonstop booze and be fine, yet you have one glass of wine and you are gone.
How much Testosterone you may release in response to a bolus dose of Testosterone Undecanoate versus another male individual, will vary based on a number of factors. This includes ester cleavage (hydrolysis of the ester to cleave it from the Testosterone molecule; the liberation step), rate of absorption of Testosterone, how much Sex Hormone Binding Globulin (SHBG) there is, and numerous other factors. But we can’t know these things until we’ve trialled them. So, we need to know how most people’s bodies will process the drug, on average. We can then apply pharmacokinetics to the average population to gather an idea of how a person should respond to a drug, making adjustments to the individual as necessary to achieve the expected or desired responses.
Models exist to simplify the conceptualisation of the various processes that take place in your body and its interaction with a chemical substance. For example, the mono-compartmental models and two compartmental models are models which are commonly divided into 5 compartments (phases), given as follows 1 :
- Liberation – Process of releasing a drug from its pharmaceutical formulation
- Absorption – The process of the substance entering the blood circulation
- Distribution – The dispersion of the substance throughout the fluids and tissue of the body
- Metabolism – The transformation or inactivation of the substance by the organism from parent compounds to daughter metabolites i.e. conversion of ethanol from alcohol into acetyl-CoA within the liver
- Excretion – The removal of the substance from the body
Using mathematical modelling techniques for predicting the absorption, distribution, metabolism and excretion of synthetic or natural chemical substances in our bodies, we can create pharmacokinetic models. Often the output will be in a graph format, as we are showing what the body is doing to a drug over time.
We appreciate that maths is not everyone’s forte, but there are a few important pharmacokinetic metrics that we want to introduce to you, all of which are within the scope of this article. This includes the following 2:
- Dose – The amount of drug administered, denoted by the symbol D
- Dosing Interval – The time between drug dose administrations, denoted by the symbol T
- Cmax – The peak plasma concentration of a drug after administration
- tmax – The time it takes to reach Cmax
- Cmin – The lowest (trough) concentration that a drug reaches before the next dose is administered
- Volume of Distribution – The volume in which a drug is distributed (varies) denoted by symbol Vd – calculated by the Dose divided by the initial concentration
- Concentration – The amount of drug in a given volume (generally plasma in the case of humans) denoted by symbol C0,Css – calculated by the Dose divided by the volume of distribution
- Elimination Rate Constant – The rate at which a drug is removed from the body given as ke
- Elimination half-life – The time required for the concentration of a drug to reach half its original value – calculated by the natural log (2) divided by the elimination rate constant
- Clearance – The volume of plasma clear of a drug per unit time, given by symbol CL
- Bioavailability – The systematically available fraction of a drug given by symbol f
Each drug will have its own Cmax and a different elimination rate constant, thus a different elimination half-life. This is also true of Testosterone and its different compounds, from different ester injectable drugs, to gels and more. In pharmacockinetics, it’s well regarded that there will be a point when the amount of drug consumed inwards, is the same as the amount of drug taken out of the system (you), which we refer to as the “steady state”. We want to reach a steady state to avoid large fluctuations in levels (although some leeway to mimic the natural diurnal pattern of Testosterone release is not a bad idea). This generally takes between 5 to 6 half-lives for most medication as we build up concentration of a drug 3. This of course depends on the drug preparation method and its half-life. For example, buccal (oral) administered Testosterone generally causes a peak of Testosterone levels within 10-12 hours of initial administration, and reaches steady state within 24 hours 4. But this will subsequently decline soon thereafter, and further administrations of the drug are necessary to reach sufficient steady states.
With some simple pharmacokinetic maths and some pharmacokinetic properties of a drug from a study population group, we can predict things like the daily release of a drug, on average. This of course is just a prediction, and individuals will have varying responses due to the aforementioned reasons. However it serves as an invaluable way of demonstrating what protocols and drugs suit a person best. We can plot all of this information onto a graph by computational means with some linear algebra.
This is exactly what we have done, for different protocols used in different practices, including our own.
Different Protocols Have Different Looking Outcomes
Indeed, different protocols WILL have very different outcomes. We can immediately visualise this with predictive graphs derived from pharmacokinetic models. As a little preface, these graphs are computationally made by The Men’s Health Clinic, created using Python 3.8, some modules, some pharmacokinetics and a lot of maths. The real outcomes from different protocols can vary greatly based on individual multifactorial differences, and we can only account for so much. However, these predictive model graphs serve as good starting points, and give us a very good insight into how different protocols may on average look in real life.
We have broken this down into the different protocols by sector, including the NHS. So, without further ado, let us start with the NHS.
NHS Protocols – According to BNF
The NHS predominantly prescribes 4 different types of protocols, these include Testosterone Undecanoate (Nebido), Testosterone Gel (Testogel/Tostran), Sustanon, and Testosterone Enanthate.
Testosterone Undecanoate (1000mg/4mL) aka Nebido®
Initial 1000mg starting dose, with second loading dose administered at six weeks. Testosterone levels are then measured after a further 12 weeks and injection intervals adjusted according to blood work.
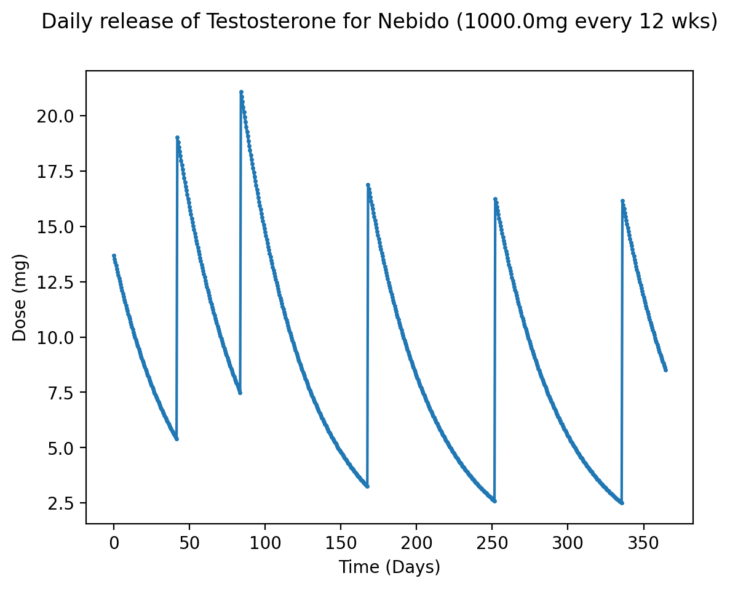
Here is what an example daily Testosterone release plot will look like for a 1000mg starting dose, with a loading dose of 1000mg at six weeks, and then injections of 1000mg every 8-12 weeks under the NHS (click on the graphs for full-size versions):
Figure 1 – Approximate daily release of Testosterone using Nebido at 1000mg every 12 weeks
As we can see, there are large swings in the daily release over time, with the Cmin becoming quite low indeed further into the year. One may experience several issues with such a protocol, including obvious fluctuations in Testosterone, uncontrolled E2, and elevated Haematocrit (HCT).
This is not to say that Nebido is not effective, nor that such a dosing pattern would not ultimately have beneficial impacts. Quite the contrary, as there is indeed an improvement in mental health and quality of life when treating men with hypogonadism via Nebido, as shown by Tong et al., 2012 5 and this was with 1000mg injections being administered at weeks 0,6,18,30, and 42. Among all participants, Testosterone levels did significantly increase to an average of 25 nmol/L by week 48, but this did take time. At week 18, Total Testosterone was still at around 15 nmol/L and unsurprisingly haematocrit increased significantly, close to being out of the upper range for men. Studies aside, clinically we will notice patients who have complained of past Nebido protocols. The manufacturers of Nebido themselves (Bayer) recommend injection intervals of 10-14 weeks, with an initial 6-week bolus dose. If one were to miss an injection by a few weeks, there could be drastic consequences, and keeping track of such long time periods can be difficult for some patients. Furthermore, every patient will have a slightly different rate of metabolism for the drug, and thus the way they respond, hence why microdosing works. It fills any gaps caused by the waiting periods between injections, preventing large peaks and troughs.
Testogel®
40.5mg applied once per day, typically in the morning. Adjustments may be made in 20.25mg increments, up to a maximum of 81mg per day. To note, one pump delivers 1.25ml of gel which contains 20.25mg of Testosterone. Note – Tostran can also be prescribed by the NHS, it is a different strength to Testogel (0.5g gel = 10mg testosterone), but the same pharmacokinetic principles apply.
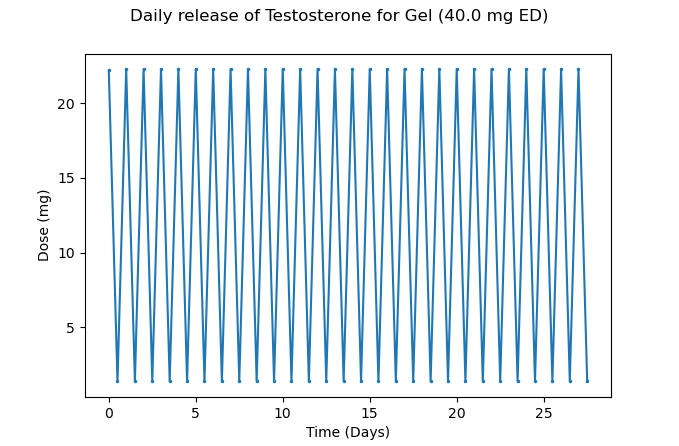
The release of Testosterone per day would look like this on a protocol of 40mg per day for one month:
Figure 2 – Approximate daily release of Testosterone using Testosterone gel at 40mg every day
Here is the maximum dose:
Figure 3 – Approximate daily release of Testosterone using Testosterone gel at 81mg every day
As you can see, there is a sharp dip towards the end of the day, not far from a baseline of 0. The reality will differ due to rate of absorption from skin to bloodstream, and some individuals will absorb the gel better than others. This may lead to unstable levels and associated issues around HCT, as well as mood and general energy levels declining towards the end of the day. Ideally, one would divide the dose and apply the gel twice per day, provided optimal absorption of the gel is achieved. This is not to say that Testosterone gel won’t work, it has already been demonstrated in studies that it does work for hypogonadal men 6, but suffice to say, it’s not optimal.
Sustanon®
The 250mg preparation of Sustanon is generally given in 1mL doses every three weeks, and then adjusted according to response.
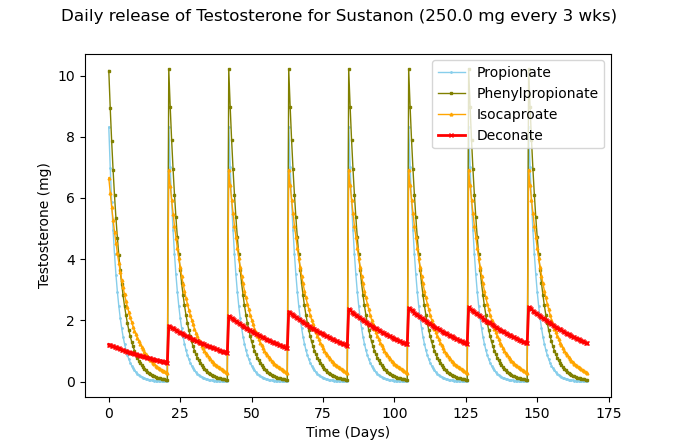
This is what the different esters look like, over a six month period:
Figure 4 – Approximate daily release of Testosterone at 250mg of Sustanon every 3 weeks with each ester highlighted in the legend
Combining the esters to achieve an average will give this:
Figure 5 – Approximate average daily release of Testosterone using Sustanon at 250mg every 3 weeks
The peaks and troughs are clearly significant, and it would take considerable time for the decanoate ester to achieve stable levels. It’s also known that this causes supraphysiological Testosterone levels, which subsequently decline very quickly and can lead to polycythaemia, a feeling of “roller-coaster” emotions, and other issues 7.
Testosterone Enanthate
250mg administered every two to three weeks, with a maintenance dose of 250mg given every three to six weeks thereafter.
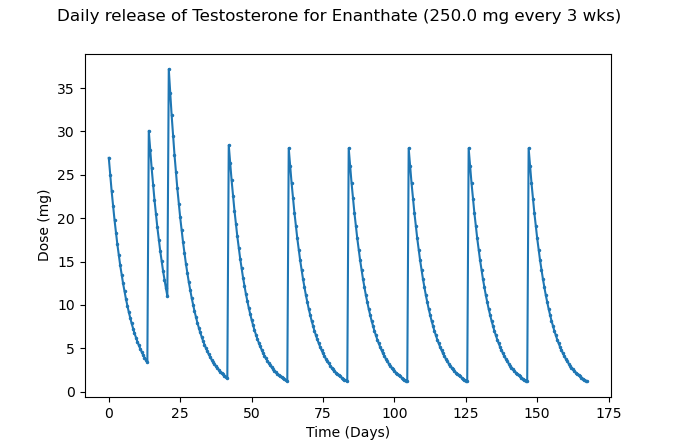
With an initial 250mg dose, followed by 250mg at week 2, and then every three weeks at 250mg, we will see this:
Figure 6 – Approximate daily release of Testosterone at 250mg of TE every 3 weeks
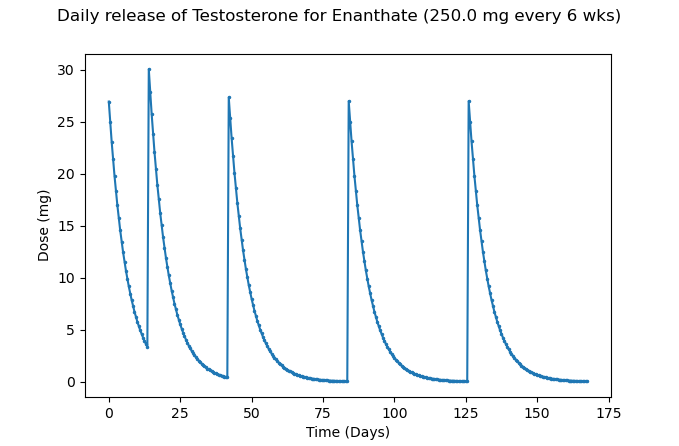
As we can see, the initial loading period does not help much in the long-term and causes even greater levels of supraphysiological Testosterone levels than Sustanon, followed by subsequent large decreases. A six week interval looks like this with the same loading phase:
Figure 7 – Approximate daily release of Testosterone at 250mg of TE every 6 weeks
This leads to significantly longer periods of low Testosterone; this will more than likely increase the risk of polycythaemia and cause a roller coaster of emotions. This is FAR from optimal, and in our opinion, this method of prescribing Testosterone enanthate should stop immediately.
Common Private TRT Clinical Protocols
Private practice allows the prescribing clinician to offer a more personalised approach to care, enhancing the patient experience. Hopefully, private practice is more than simply a fast-track service to treatment and a nice cup of coffee on arrival at a plush clinic! NHS protocols are safe, but they tend not to allow for prescribing outside of the guidelines. However, it is possible to prescribe medications in an ‘off-label’ fashion. This simply means that the medicine is being used in a way that is different to that described in the product licence, which is agreed by the Medicines Healthcare Regulatory Authority (MHRA). It is even possible to prescribe medications for an intended purpose when there is no product license, such as Transscrotal Testosterone Cream, but this must be done with extreme caution. The prescribing clinician must have sufficient knowledge and expertise to prescribe said medications, and have made the patient fully aware of the rationale behind why they have prescribed medications in this manner and ensured that they are happy with this methodology. ‘Primum non nocere.’ In the private medical sector, practices vary widely, from doctor-led clinics to online providers, generally trending towards increased frequency protocols to help achieve more stable pharmacological levels. Though far from perfect, this generally leads to better outcomes than traditional NHS protocols. Let us examine it in further detail.
Sustanon®
125mg administered every five days via deep intramuscular (IM) injection. It is important to note that a deep intramuscular injection will generally lead to a faster rate of absorption, but also a subsequent quicker decline in Testosterone levels too.
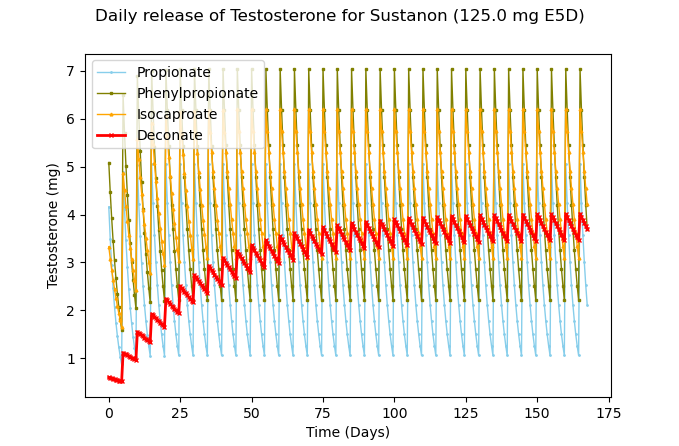
For each individual ester, this is what the daily release would look like:
Figure 8 – Approximate daily release of Testosterone at 125mg Sustanon every 5 days with each ester highlighted in the legend
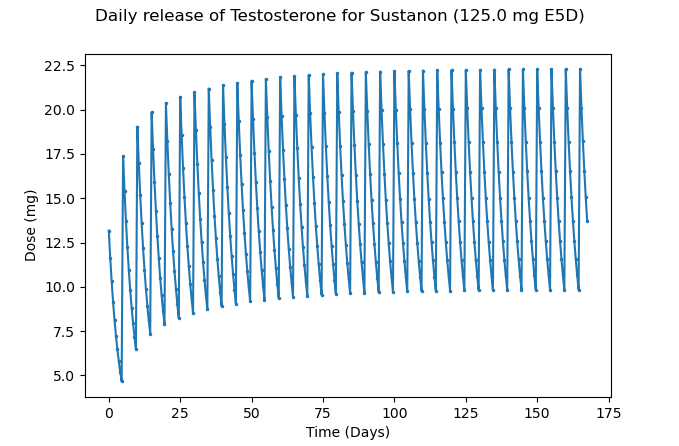
And integrating the graph for an average release, we get this:
Figure 9 – Approximate average daily release of Testosterone at 125mg of Sustanon every 5 days
This is much better than the NHS prescribed protocols, and despite still seeing large peaks and troughs, one can expect to experience less issues with a protocol prescribed like this, as well as improved outcomes on biochemical assays, mental day to day wellbeing, energy levels, symptom resolution and a generally improved quality of life.
Testosterone Enanthate
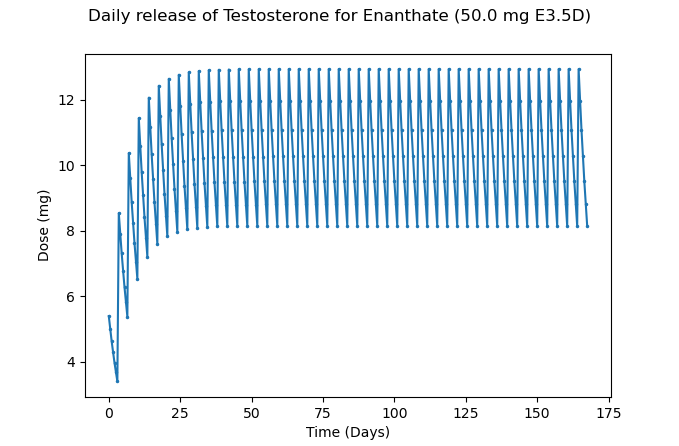
50mg administered every 3.5 days via shallow IM injection.
Figure 10 – Approximate average daily release of Testosterone at 50mg of TE every 3.5 days
As we can see, more stable levels are reached with smaller differences between the peaks and troughs. Still, the difference between the peaks and troughs could be reduced further, which ultimately leads to greater improved patient outcomes.
Trans-scrotal Testosterone Cream (100mg)
Cream applied every 12 hours.
Trans-scrotal application of Testosterone cream is becoming increasingly popular. Scrotal skin does indeed have the thinnest stratum corneum (outer layer of skin) and high steroid permeability, whilst minimising the risk of passive transfer to others 8. Although this significantly elevates DHT due to increased enzyme concentrations of 5α-reductase in the testes. Studies do show generally good levels of success with this method, as shown by Lyer et al., 2017 in the below figure for application of 50mg, 25mg, and 12.5mg:
Figure 11 – Administration of Trans-scrotal cream at different doses and serum Total Testosterone 8
Unfortunately this is not always the case for every man. Some may struggle due to excess hair, excess conversion of Testosterone to DHT or general poor absorption. Furthermore, as one can clearly see, 100mg would cause potentially supraphysiological levels on average, as 50mg elicits as high as 680ng/dL, or 23.6 nmol/L. Application within 12 hours would then cause another subsequent peak that gradually increases levels. Ideally, one would use a lower or mid dose cream and apply twice per day until stable levels are met. This is not to discredit the cream, as it works for some males and it can indeed mimic natural diurnal Testosterone release well too.
Human Chorionic Gonadotropin (HCG)
350-500iu administered every 3.5 days, or 500iu three times per week.
HCG is a necessary component for successful TRT outcomes. It stimulates the testis, even to a small degree, which helps to produce hormones other than just Testosterone and Oestradiol. Hormones that are are important to your health and wellbeing. In the case of physical castrates, HCG may have beneficial effects elsewhere in other tissue such as the brain; elevated brain LH may well improve general brain health and prevent diseases such as Alzheimer’s disease 9. Therefore, we can presume that if HCG crosses the blood-brain barrier, we may observe similar effects 10. The dose would of course vary for someone who is physically castrate, most likely being lower. Therefore, do not discredit it – the NHS would be well advised to begin including HCG into their Testosterone therapy treatment regimes, too.
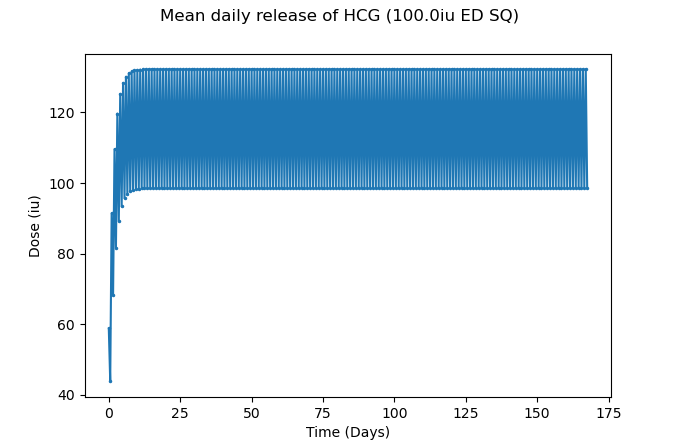
At a release dose of 500 iu every 3 days, via a subcutaneous route, we would see something like this:
Figure 12 – Approximate average daily release of HCG at 500iu every 3.5 days, via subcutaneous administration
It should be noted that this does not show the biphasic nature of HCG, where a subsequent peak will occur. Such a dosing protocol may over time lead to excess oestradiol due to large peaks and troughs. With large peaks and troughs, there may again be more supraphysiological responses and quick declines, ergo our previous reasoning for not wanting this.
At 350iu, we will see the exact same graph, but with a smaller Y axis; i.e. the dose is smaller as we use a smaller dose to begin with. Again, this does not include obvious variations between patients, such as absorption rate due to varying blood flow at the site of fat.
The Men’s Health Clinic – Typical Starting Protocols
At The Men’s Health Clinic, we encourage microdosing. Using the most effective injection frequency with the lowest viable dose, to achieve the most optimal patient outcome. This includes looking at patient symptoms, discussion of how the patient feels, biochemical assays/profiles, body composition changes, energy, activity performance and mental wellbeing. We adjust the dose based on these parameters, and include ancillaries such as aromatase inhibitors where necessary and as agreed with the patient.
Testosterone Cypionate
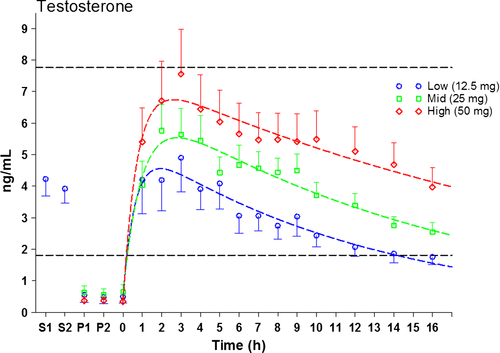
12.5mg administered daily via subcutaneous injection.
Dosing via a subcutaneous route will lead to altered pharmacokinetics, such that a more stable release of the drug is administered due to reduced blood flow in the fat tissue. Combine this with microdosing more frequently, and we will see much smaller differences between the peak and the trough, leading to far more stable daily levels of Testosterone, as shown below:
Figure 13 – Approximate average daily release of Testosterone at 12.5mg of TC every day
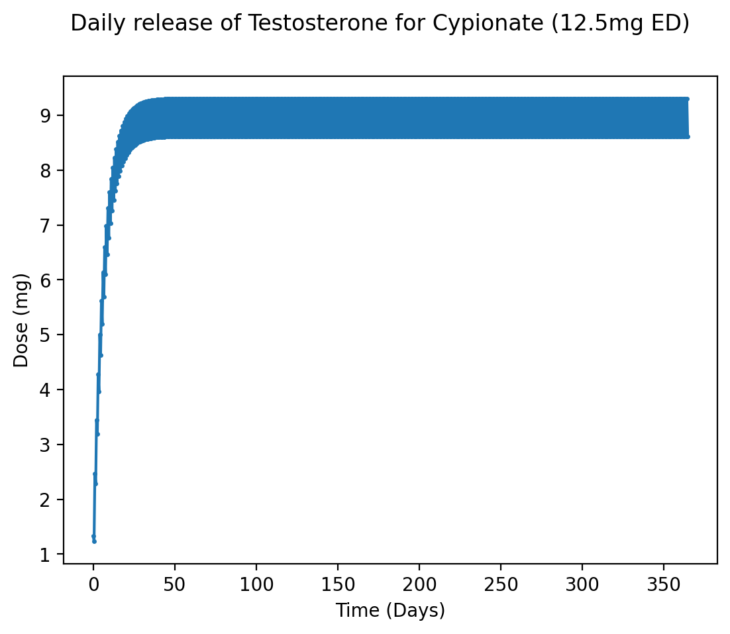
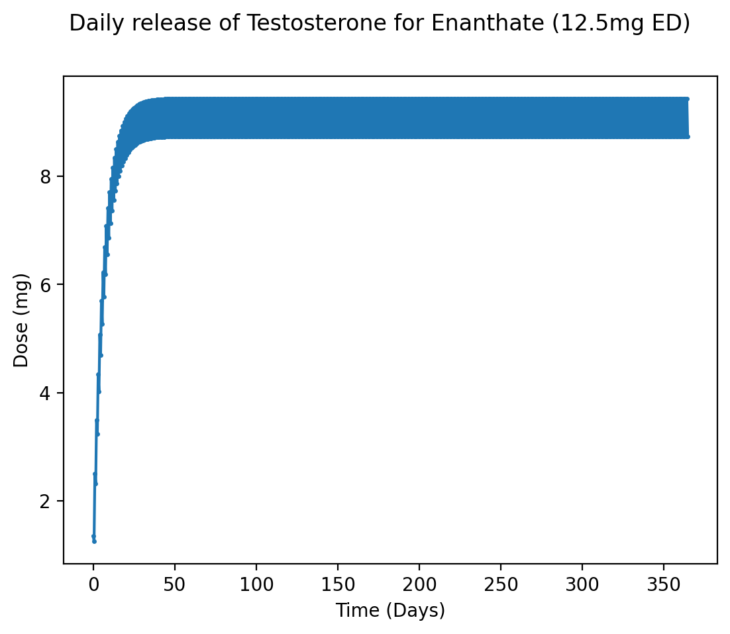
Testosterone Enanthate
12.5mg administered daily via shallow IM injection.
With Testosterone Enanthate we see a very similar pattern, even with IM injections, though there is a slightly higher difference between the peak and trough due to a small half-life variation with Testosterone Enanthate and Testosterone Cypionate:
Figure 14 – Approximate average daily release of Testosterone at 12.5mg of TE every day
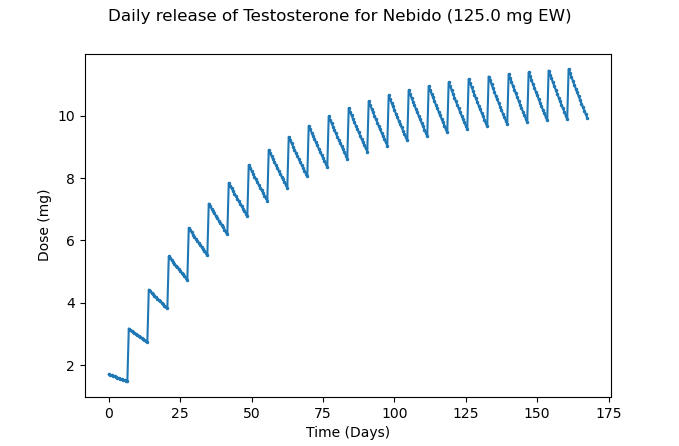
Testosterone Undecanoate (1000mg/4ml) aka Nebido®
125mg (0.5ml) administered weekly via deep IM injection.
Nebido takes a significant amount of time to reach stable levels due to its relatively long half-life, but at a dose of 125mg every week, we do ultimately reach stable levels with relatively small fluctuations between the peak and trough for daily release as shown below:
Figure 15 – Approximate average daily release of Testosterone at 125mg of Nebido every week
If this methodology was to be adopted by the NHS or within private practice, a loading dose regime could improve the time taken to achieve stability.
At 250mg every two weeks, there is greater variation again. While this is to be expected, it is still superior to other protocols (including the manufacturer’s recommended one), as we do not reach supraphysiological levels, nor do we hit a ‘rock-bottom’ and return to near hypogonadal levels prior to the next injection. This ensures optimal hormonal balance, health, wellbeing, and quality of life, whilst taking fewer injections.
Human Chorionic Gonadotropin (HCG)
100iu administered daily via subcutaneous injection.
HCG, much like Testosterone Cypionate, is typically painless to inject daily. There is little reason to not be doing this and I have already given reasons for why you should potentially be taking it even if you are physically castrated! So, do not discredit it. Stability with HCG is also warranted so we do not cause spikes in E2 and other associated issues with large spikes in Testosterone and Oestradiol. It will look like this:
Figure 16 – Approximate average daily release of HCG at 100iu every day, via subcutaneous administration
As you can see, there is at most a ~30iu difference from the peak and trough, much better than the near 200iu difference seen in private practices elsewhere. This will lead to more stable levels and less E2 spikes, ergo fewer overall issues.
Our Patient Zero: The Optimal Male & Evolution of Practice at The Men’s Health Clinic
We have discussed the evolution of our clinical practice in ‘Gold Standard TRT’. Whist the BSSM guidelines have provided us with a safe platform to work from, we believe that our current patient model is far more progressive and serves in the best interests of the individual. Medicine is a practice; a progressive clinician will continually seek to improve the level of care that they offer to their patient. It is safe to say that times have changed, our understanding of Testosterone Deficiency and TRT has increased exponentially and we have made the necessary adaptations. We have faced numerous challenges along the way, and we are certain that we will face many more. However, we have approached each one as an opportunity to either consolidate, learn, or improve. Dogma should have no place in medicine. With that, all of our patients, even those that have been on their TRT journey from the inception of the clinic, are now microdosing, as we are confident that it is the most effective way of achieving stable male androgen levels. We would like to thank them for their patience and understanding.
Our Patient Zero is still with us today and has agreed to let us use his data to demonstrate on both an anecdotal and case study level, how effective microdosing has been for him. He is a 57-year-old male, who presented with Testosterone Deficiency. He demonstrated clinical symptoms of hypogonadism, in addition to having concerns about loss of concentration, memory issues and weight gain.
In January 2016, we commenced his first trial of TRT with Testosterone Undecanoate, the UK licensed drug for Testosterone Deficiency. This began with an initial saturation dose of 1000mg, followed by a second six-week loading dose of 1000mg, and then follow up doses of 1000mg every 12 weeks. After a suboptimal trough reading of 13.6nmol/l in September 2016, eight months after starting TRT, we made the decision to switch him to a 10-weekly injection interval. In December 2016, we also made the decision to introduce 500iu of HCG every 3.5 days because, despite shortening his injection interval and him noting a minor improvement in his sense of well-being on TRT, he still felt flat. Disappointingly, Patient Zero’s trough levels remained suboptimal so in January 2017, we moved him to shorter eight-week injection intervals. He had subsequent issues with elevated oestradiol and so Anastrozole (an aromatase inhibitor) was commenced to manage this. Disappointingly, after 15 months on Testosterone Undecanoate, alongside 300iu of HCG and 0.5mg of Anastrozole every 3.5 days, his trough level was still only 15.1nmol/l.
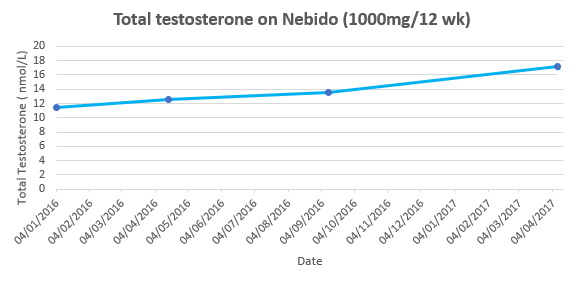
When plotted on a graph Patient Zero’s Testosterone levels whilst on Nebido from commencement of treatment looked like this:
Figure 17 – Total Testosterone levels of patient Zero (NHS/TMHC) on Nebido, 1000mg/12 wks
In April 2017, we made the decision to switch him to Testosterone Enanthate, a shorter-acting ester, so that we could more effectively titrate his dose according to response. As with all medical protocols, there is no one size fits all methodology and Patient Zero did not fare well with every 3.5-day intramuscular injections of Testosterone Enanthate. He unfortunately suffered issues with elevated oestradiol and haematocrit, necessitating intervention. We subsequently made the decision to stop HCG, as the logistics of keeping the medication refrigerated made compliance an issue, and the hormonal fluctuations would have been problematic.
Despite the rocky start to therapy, I am pleased to report that Patient Zero has been on daily subcutaneous injections of Testosterone Cypionate (17.5mg per day), with 6.25mg of Exemestane (an aromatise inhibitor) every other day since November 2019, and is faring much better. He now has optimal male androgen levels – Total Testosterone 28.2nmol/l and Oestradiol 97pmol/l – all other biomarkers are healthy and he feels a subjective improvement in his overall sense of well-being. A graph will follow once we’ve gathered enough data points to demonstrate this.
Case Study 2
Another patient, a 37-year-old male, also joined our clinic during our initial TRT evolution. He too was commenced on Testosterone Undecanoate, however due to his age, he was also started on 500iu HCG every 3.5 days to help preserve fertility. Again, with an initial saturation dose of 1000mg Testosterone Undecanoate, followed by a six-week loading dose at 1000mg and then a follow up dose at 12 weeks. His injection interval was then adjusted according to his qualitative symptoms and trough readings.
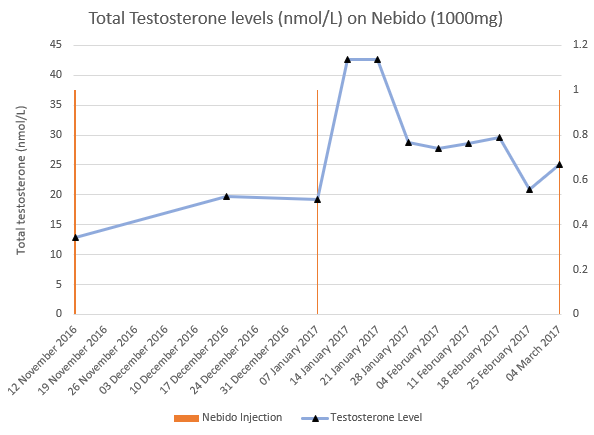
We were initially reassured by his healthy Testosterone levels. The stark contrast in his levels with patient zero is in part due to the HCG causing production of intra-testicular Testosterone. Despite this, he unfortunately described a rollercoaster of emotions the further away he was from his last injection, something that was becoming more commonly reported amongst our patients on Testosterone Undecanoate. We elected to measure his Testosterone levels on a weekly basis to see if we could demonstrate a quantitative change in his levels, to support his subjective symptoms. The results are demonstrated in the graph below:
Figure 18 – Total Testosterone levels of another patient (TMHC) on Nebido, 1000mg. The orange bar represents injection points; the blue line represents Total Testosterone with black triangles showing data points
As you can see, despite having healthy trough levels, there was significant variance in his Testosterone level between injections, providing false reassurance of stability. This graph clearly demonstrates the ‘peak and trough’ that is often described by patients on such wide injection intervals. As mentioned before, we should be looking to achieve stable levels. Likewise, there is wide variation in both practice and standards within the private domain. TRT is a complex medical condition that, without exception, should always be doctor-led. A doctor who specialises in, and understands the nuances of, TRT will always achieve healthier outcomes than patients whose condition is managed by a non-medical “case manager”. Just as importantly, ensure that you find a doctor who is open to progressing with the latest methods because, whilst patients should have realistic expectations, they have every right to expect the best possible treatment, especially if it comes at a cost.
Discussion & Conclusion
Male androgen levels follow a diurnal pattern, with anabolic processes predominating during the night, meaning that a man’s natural levels are highest early in the morning. This is the reason that Testosterone levels are ideally measured between the morning hours of 08:00-11:00, in a fasted state as digestion lowers Testosterone. A clinician would need to gauge how effective the Hypothalamus Pituitary Gonadal axis (HPGA) is at producing serum Testosterone before other physiological processes, such as digestion and physical exercise, alter these serum Testosterone levels. This is important as it provides a level of consistency for statistical comparison, and it is well-recognised that patients on TRT should have their levels measured in a trough for the very same reason.
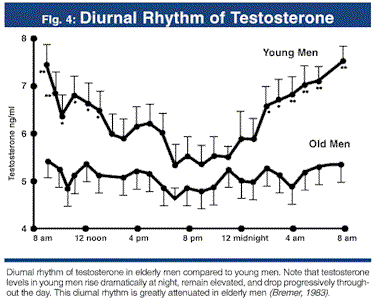
The 24-hour diurnal rhythm of natural testosterone production is demonstrated in the graph below. Whilst there is variance over the course of a day, on average the typical healthy and natural male will produce between 6 to 10 mg of testosterone per day, highlighting that there is always relative stability over the course of 24 hours 11,12. You will note that the variation in Testosterone is relatively minor in young men, while seemingly less present in older men, this is likely due to a relative increase in SHBG that occurs with advanced age concurrent with a decline in Lutenising Hormone (LH) output and reduced testicular response – this is briefly discussed in our ‘TRT, SHBG & Health – Facts, Questions and Evolution‘ blog.
Daily microdosing of Testosterone Cypionate (or Enanthate) and HCG, offers an effective method to mimic this natural physiological production. This is because it will still cause a peak and trough to be produced, but with a small variation between the peak and trough once stable levels are reached, much like what’s seen in the natural diurnal rhythm of Testosterone among young men (Figure 19), and what has been demonstrated in the above graphs. This is in stark contrast to protocols which advocate longer injection intervals, which ultimately lead to far greater variations in peaks and troughs. This will usually cause extreme changes in levels that are not seen in the daily variation of Testosterone, nor are they seen even in longer time periods.
Figure 19 – Natural testosterone production in a 24 hour time period demonstrating diurnal rhythm in young and older men (Bremner WJ et al., 1983)
In clinical practice, the difference that daily injections of Testosterone Cypionate (or Enanthate) have had on stability has also been quite marked. Our patients who have transferred from relative stability on less frequent injection protocols have all, without exception, noted a qualitative difference in their sense of wellbeing now that they are microdosing and would not choose to go back to their previous injection regimen.
The quantitative level of a particular hormone present in your body at any one time has a direct and indirect effect on numerous physiological processes. To achieve homeostasis, too much is often as bad as too little in what is a very complex mechanism that necessitates fine control for optimum function. With microdosing, we have noted fewer clinical side effects, such as raised haematocrit and elevated Oestradiol which are well recognised with other protocols. Once clinical practice has returned to normal after the COVID-19 pandemic, we plan to engage in a clinical trial to demonstrate the effectiveness of microdosing.
Interestingly, we have also noted improved compliance with small daily dosing into the subcutaneous tissue, as injections are relatively painless compared to the more traditional intramuscular injections. Daily administration also means that injecting simply becomes a part of your morning routine, much like brushing your teeth.
“Although the thought of every day injections was daunting to start with, the positive effects it had on my well-being, far outweighs the downside of having to inject daily.” – Alex, 38.
The aim of TRT should be to restore Testosterone levels and their metabolites – Oestradiol and Dihydrogentestosterone (DHT) to within normal physiological parameters. This helps to facilitate growth and repair and improves mental wellbeing, in addition to metabolic health and sexual function. A basic goal should be to reverse the confirmed Testosterone deficiency. From a patient perspective, the primary goal might be to reverse the negative symptoms of low Testosterone that they initially presented with. The BSSM guidelines suggest aiming for a Total Testosterone of between 15-30 nmol/L. Disappointingly, this does not consider the effect of SHBG and Albumin on Free Testosterone, Oestradiol, and DHT. The importance of SHBG has been discussed in ‘TRT, SHBG & Health – Facts, Questions and Evolution‘. We need to adopt a holistic approach to patient care as both qualitative well-being and quantitative markers can differ widely, between 15 nmol/L and 30 nmol/L, based on the individual.
From analysing and reflecting on the above data, it should now be clear why we consider gold standard TRT to be DAILY Testosterone and HCG injections. The goal of TRT is to provide the necessary foundations for achieving hormonal balance and attaining a semblance of normality in one’s life.
We believe clinicians should adopt a more progressive approach to TRT. The aim should be to both normalise levels, help maintain function and reverse any negative symptoms. TRT should be considered Hormone Replacement Therapy. It is well-recognised that exogenous Testosterone suppresses both the release of both LH and Follicle Stimulating Hormone (FSH) from the pituitary gland. This means that LH no longer stimulates the Leydig cells of the testes to produce Testosterone, and correspondingly, FSH no longer stimulates the Sertoli cells to produce sperm. This is a simplistic statement, as there are other complexities involved, such as aromatisation of Testosterone to Oestradiol within the testes to help facilitate spermatogenesis.
Human Chorionic Gonadotropin (HCG) mimics LH, and is used by the NHS to treat male infertility. We use HCG with TRT as it provides stable drug levels, due to its relatively long half-life compared to that of LH. However, LH is released from the pituitary gland as a response to stimulation from Gonadotropin Releasing Hormone from the hypothalamus, which itself is released in a pulsatile manner. As already stated, anabolic processes predominate at night (or during rest periods), ergo there is not constant stimulation of the Leydig cells to produce Testosterone and subsequent Oestradiol within the testes. Whilst HCG binds to the same receptor as LH, its mechanism of action and therefore effects are more constant. Using an LH-analogue would clearly be impractical. The constant activation of the Leydig cells means a slightly suboptimal dose is required due to otherwise excess aromatisation of Testosterone to Oestradiol
Disappointingly, neither HCG nor Oestradiol are mentioned within the BSSM guidelines. The topic of Oestradiol deserves its own article as it has numerous health benefits. Academically, there is an optimal ratio of Testosterone to Oestradiol, something discussed in ‘The Importance of Oestrogen in TRT’. However, as with most things in biology, principles are not the same as rules, biological individuality sometimes confirms this. This is especially true regarding an optimal Oestradiol level and the T:E ratio.
Traditional TRT is much like the old Ronseal advert “it does exactly what it says on the tin”, however, as already discussed we prefer the idea of maintaining function with HCG, whilst optimising Testosterone levels with Testosterone. While the primary role of LH/HCG is to produce intra-testicular Testosterone and improve fertility (we have had over 30 successful conceptions amongst our patient cohort), the role of LH extends beyond this.
It must be noted that HCG should only be prescribed under expert supervision, as it does not follow a dose-dependent effect on Testosterone levels. Men present with varying degrees of primary (testicular), secondary (brain) and/or tertiary (prolactin) hypogonadism. Very few patients are solely primary, unless of course they are castrates, and no one is solely secondary (except in very rare diseases), so the effect of a fixed dose of HCG needs to be witnessed in clinical practice and the dose of Testosterone adjusted accordingly. Some of my younger patients have had their starting dose of Testosterone Cypionate carefully titrated down to just 5mg daily (3.5mg if you take out the ester attached to the Testosterone molecule), in order to keep them within normal physiological parameters and optimal well-being. Again, as a point of reference, the average healthy young male produces approximately 6mg of Testosterone per day, with concentrations as high as 10mg per day 11,12. As we’ve said numerous times before, more is not better, both objectively when assessing biomarkers and subjectively, when describing qualitative well-being. We are merely seeking normality.
The premise behind TRT is achieving stable male androgen levels to help facilitate anabolic processes. Testosterone, Oestradiol, and DHT are all involved in these mechanisms and there is an optimal quantitative number for each parameter, the ratio of T:E:DHT appears as important as the absolute number. Our biological individuality means that a patient’s dose and ratio must be titrated according to affect. Whilst some patients may initially seek high-normal levels, in practice we have lowered the dose of Testosterone in numerous men already on TRT to a more mean figure, only for them to report a subsequent improvement in their libidos and a lessening of previously reported anxiety or heightened state.
Gold standard TRT extends even further than this, the aim is to also optimise function by giving men an appropriate dose of Testosterone and HCG for their genetics, physiological requirements, and subsequent utilisation. This can only be done through careful titration, its certainly a case of “slow and steady wins the race”. Administration of a medication forces a physiological reaction, even if that medication is “body-identical”, a new buzz term in the healthcare sector. This means it has a subsequent knock on effect on all the parameters that Testosterone, Oestradiol, and DHT influence. SHBG is a perfect example of this. It’s role is discussed in detail in ‘TRT, SHBG & Health – Facts, Questions and Evolution‘. Simply put, it is a ‘buffer’ for Testosterone, Oestradiol. and DHT; a rise in SHBG appears to confer a positive health advantage. Aggressive doses and erratic protocols appear to lower SHBG, which is one of the reasons we are not proponents of using high dose transscrotal testosterone cream. We have seen a reassuring rise or normalisation in SHBG with microdosing. Paradoxically, I am often reassured when a previously stable patient reports that their sense of well-being has declined very slightly, as the blood parameters often demonstrate a slight rise in SHBG. A slight tweak to their protocol ensures the return to qualitative wellbeing, alongside improved health.
There are complexities in ascribing what is a ‘normal’ physiological range, something discussed in detail in ‘Testosterone Reference Ranges – What Should Be Considered “Normal” & Why?‘. The fact that reference ranges are consistently lowering, should strike alarm in both clinician and patient. Applying normal to a sick population is not acting in the best interests of the patient, this drop in the new normal cannot reflect evolutionary change. There are countless contributory elements including ‘Endocrine Disruptors & The HPG Axis‘. However, we believe obesity and lack of physical activity are the largest contributory factors resulting in a decline in Testosterone levels, discussed in depth in ‘Me & My Fat – The Role of Fat on Your Hormonal Health‘. The aromatase enzyme, which converts Testosterone to Oestradiol is located in both peripheral and visceral adiposity. This is the biggest single challenge for both patient and clinician in attaining a healthy Testosterone to Oestradiol ratio for optimal well-being.
We firmly believe that Testosterone Deficiency and Testosterone Replacement Therapy needs a champion in the UK. Multiple studies and research have already demonstrated TRT to be safe, but it needs to be more effective. We need to challenge current attitudes and practices to improve patient outcomes. We should be responsive to both qualitative and quantitative research, alongside clinical experience. We need to listen to the patient but retain a necessary element of objectivity so that we always act in their best interests. Whilst a progressive approach may imply an added level of complexity, in fact the contrary applies. Keep it simple, keep it safe. We need a more logical, common sense approach to TRT. If we apply our current knowledge and understanding of pharmacokinetics to the available medications; smaller, more frequent injections will lead to more stable male androgen levels. This is certainly what we have found in our own clinical practice.
It has been a fascinating, yet challenging journey to date, we look to the future with aspirations of changing practice. With that said, gold standard TRT is microdosing daily subcutaneous Testosterone Cypionate (or Enanthate) and HCG injections.
Dr Robert Stevens MBChB MRCGP Dip.FIPT
Joseph Hearnshaw BSc (Hons) DPS, MSc, MRSB
References
- Rxkinetics. Section 1 – LADME Scheme. http://www.rxkinetics.com/pktutorial/1_2.html. Accessed August 9, 2020.
- AGAH working group PHARMACOKINETICS. Collection of Terms, Symbols, Equations, and Explanations of Common Pharmacokinetic and Pharmacodynamic Parameters and Some Statistical Functions .; 2004. https://web.archive.org/web/20160508185420/http://www.agah.eu/fileadmin/_migrated/content_uploads/PK-glossary_PK_working_group_2004.pdf. Accessed August 9, 2020.
- Thomas A. M. Kramer. Side Effects and Therapeutic Effects. Medscape General Medicine. https://www.medscape.com/viewarticle/448250_3#:~:text=We call this %22steady state,time to reach steady state. Published 2003. Accessed August 18, 2020.
- Shoskes JJ, Wilson MK, Spinner ML. Pharmacology of testosterone replacement therapy preparations. Transl Androl Urol. 2016;5(6):834-843. doi:10.21037/tau.2016.07.10
- Tong SF, Ng CJ, Lee BC, et al. Effect of long-acting testosterone undecanoate treatment on quality of life in men with testosterone deficiency syndrome: A double blind randomized controlled trial. Asian J Androl. 2012;14(4):604-611. doi:10.1038/aja.2011.178
- Wang C, Cunningham G, Dobs A, et al. Long-Term Testosterone Gel (AndroGel) Treatment Maintains Beneficial Effects on Sexual Function and Mood, Lean and Fat Mass, and Bone Mineral Density in Hypogonadal Men. J Clin Endocrinol Metab. 2004;89(5):2085-2098. doi:10.1210/jc.2003-032006
- Yassin AA, Haffejee M. Testosterone depot injection in male hypogonadism: a critical appraisal. Clin Interv Aging. 2007;2(4):577-590. http://www.ncbi.nlm.nih.gov/pubmed/18225458. Accessed August 15, 2020.
- Iyer R, Mok SF, Savkovic S, et al. Pharmacokinetics of testosterone cream applied to scrotal skin. Andrology. 2017;5(4):725-731. doi:10.1111/andr.12357
- Blair JA, Bhatta S, McGee H, Casadesus G. Luteinizing hormone: Evidence for direct action in the CNS. Horm Behav. 2015;76:57-62. doi:10.1016/j.yhbeh.2015.06.020
- Movsas TZ, Weiner RL, Greenberg MB, Holtzman DM, Galindo R. Pretreatment with human chorionic gonadotropin Protects the neonatal Brain against the effects of hypoxic-ischemic injury. Front Pediatr. 2017;5:3. doi:10.3389/fped.2017.00232
- Testosterone, aging, and the mind – Harvard Health. https://www.health.harvard.edu/newsletter_article/testosterone_aging_and_the_mind. Accessed August 19, 2020.
- Handelsman DJ, Conway AJ, Howe CJ, Turner L, Mackey M-A. Establishing the Minimum Effective Dose and Additive Effects of Depot Progestin in Suppression of Human Spermatogenesis by a Testosterone Depot*. Vol 0.; 1996. https://academic.oup.com/jcem/article/81/11/4113/2649578. Accessed August 21, 2020.