Endocrine Disruptors & The HPG Axis

Plastics, soya milk, heavy metals and many other compounds have been the subject of scrutiny in the past as ‘endocrine-disrupting chemicals’ (EDC). In this blog I’m going to try separate fact from fiction, giving you the science and talking about what potential, or known, EDC’s exist, and how they affect your endocrine system.
What is an Endocrine-Disrupting Chemical (EDC)?
EDCs are exogenous compounds that can interfere with any aspects of endogenous hormone production; release, transport, metabolism, binding, action, or elimination 1,2. This can include pesticides, fungicides, plastics, synthetic chemicals that are used as industrial solvents, heavy metals, and various pharmaceutical agents that can sometimes be found in water supplies. Some EDCs are even naturally occurring and found in our diet. We can find EDCs in a variety of everyday products, including your clothes, toothpaste, soap, carpet, and paper 3.
There exists an EDC database, The Endocrine Disruption Exchange, that lists all the EDCs. It was last updated in 2018 with 1482 EDCs being recorded 4. We are continuously exposed to various EDCs via food, water, air, and consumer products. It is important to note that our understanding and knowledge of EDCs is limited. There is continued debate on what effects EDCs can have on us. For example, reviews on the effects of bisphenol A (BPA – commonly found in plastics) in humans has had conflicting results reported regarding its effects of pubertal development and diabetes 5,6.
I want to first make it very clear that there are complications of determining what effects EDCs have in the real world on humans. For one, many studies use a mixture of EDCs; this contains simple and unrealistic mixtures compared to real world EDC mixtures. There are other complex reasons, so don’t take what is said as an end statement to say, ‘stop using plastic because of BPAs’ (there are other very good reasons to avoid or instead recycle plastics, though) or ‘stop drinking soya milk because the phytoestrogens will grow my breast tissue!’. Not until we can ascertain what real world EDCs do to us, and scientific consensus is met 7.
But, let’s review a few EDCs that may affect the Hypothalamic-Pituitary-Gonadal Axis (HPGA) and thus hormones of the HPGA such as Testosterone.
EDCs and HPGA hormones
Some studies state that over the past decades, male reproductive health has been on the decline, with an increase incidence of hypogonadism. Concurrent with this is an increase in the number of EDC levels. From this, there is a suggested argument that EDC exposure will contribute to increased risk of male infertility and/or hypogonadism 3. However, assumptions based purely upon correlation are not to be equated with causation; that is to say that this doesn’t mean it’s true that EDCs present in our environment are causing more low Testosterone incidents. The answer is that we do not know for certain. It is difficult to ascertain whether such relationship exists in the real world to innumerable factors, including the concentration of EDCs, different types present, and other uncontrolled environmental and genetic factors. Furthermore, many of the human studies don’t report key information such as body composition, which of course is a cause of hypogonadism and worsened fertility. There are also varying metabolic rates among individual humans (people), which can account for very different responses to EDC exposure 3; what may make your Testosterone drop, may not affect another man! So again, take what is said with a healthy dose of critical thinking; it may not translate to real world results.
It is undeniable though that we are identifying EDCs in the urine of subjects, where such identifications were not found in the past. For example, BPA has been detected in at least 90% of subjects in the USA, Germany, and Canada. Phthalate metabolites (used to soften PVC polymers) have also been identified in 75% of Americans. We know that both phthalates and BPA can negatively affect male reproductive health in animal and cell studies 8,9. Several rodent studies demonstrate lowered testosterone with certain phthalates, such as DBP. One may argue that rats don’t represent good human models, indeed they are not humans, but even Faure et al., 2013 identified that exposure of dioxins in men caused more abnormal sperm, and Jurewicz et al., similarly reported decreased sperm quality with polychlorinated biphenyl (PBC) 10,11. PCBs are notable for blocking normal thyroid system function and we most often derive them from food, due to bioaccumulation (i.e. fish eats plastic from polluted river, bigger fish eats fish, then you or another animal eats the fish, or it ultimately ends up in other food stock, albeit in smaller concentrations).
Just to clarify a bit of terminology, phthalates are known as plasticisers, which are added to plastics to increase their flexibility, transparency, durability, and longevity. They are often used in PVC. Dioxins are a group of chemical compounds, which are also persistent environmental pollutants and are often toxic. PCBs are included among this grouped list, and they tend to accumulate in our food chain – sources include uncontrolled combustion (i.e. wildfires/accidental fires), metal smelting, engines using leaded fuel containing certain additives, and certain pesticides. To note, there are no natural sources of PCBs; they are man-made. But PCBs are stable and remain in the environment for a long time, and accumulate in water, the biosphere (all the ecosystems), and small amounts in the air. This a by-product of man’s excessive use of PCBs and improper disposal, going as far back as its use in the 1930s in the electric industry 12.
For those old of us to remember the Vietnam war and the Malayan Emergency, Agent Orange handling would cause high exposure of dioxins. Sadly, PCBs are often known to flush from industry and land to contaminate fish, but thankfully we try meet a minimum food safety standard and this is less of a problem in the UK and EU. This should illustrate to you how easy it is for you to be exposed to different EDC’s; from food, to fires, to burned PVC piping! But plastics are becoming BPA-free more often and I should hope none of the readers amongst us are pyromaniacs that cause uncontrolled fires and inhale the smoke directly, regardless. This does not mean you can’t have a barbecue, it’s more about avoiding inhaling the smoke from uncontrolled fires that often turn into wildfires and produce large volumes of smoke, burning various different types of materials. But it would be wise, for your own health, to avoid purposely trying to inhale as much BBQ smoke as your lungs can possibly do for prolonged periods of time.
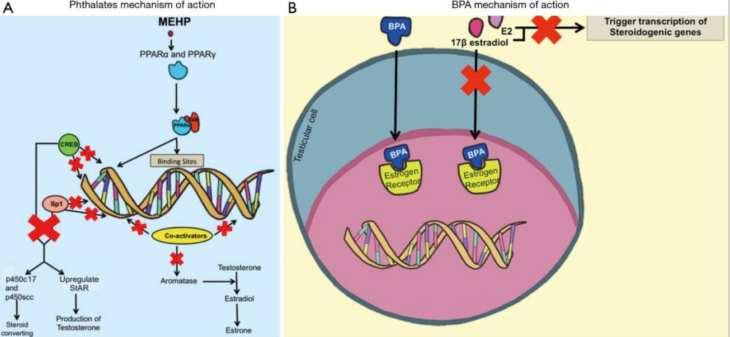
So, I have gone on about phthalates and other EDCs here without explaining about how they are lowering testosterone synthesis and reducing fertility. Well, the below figure represents it well. Figure 1 (A) is complex, so bear with me; what is essentially happening here is the Phthalate (MEHP) is blocking, what is ultimately, the production of sex-steroid hormones. In Figure 1 (B), what we see is BPA blocking the oestrogen receptor and preventing the binding of Oestradiol, thus increasing the concentration of Oestradiol, which will ultimately lead to lowered Testosterone via negative feedback. Furthermore, this ultimately leads to a direct blocking of the ‘transcription of steroid-producing genes’ (plain English: stops sex-steroid production at the gene level, too) 3. Furthermore, BPA can block androgen receptors 13–17.
Not described in this figure is the process of reducing semen quality; all you need to know is that EDC’s can do this.
Figure 1 – EDC’s mechanism of action which causes lowered sex-steroid hormones
(click for full-size image)
If you have paid attention, you know that these are only a few EDCs. What about food? There has been a lot of ‘internet broscience’ hype around soy, popularised by ‘soyboy’ ‘memes’. This has unfortunately caused a re-emergence of a notion that soya can lower Testosterone and induce gynaecomastia. What does the science say?
One study reported a 19% decrease in mean serum testosterone levels in a study with 12 men over a 4-week period, in response to consuming 56g isolated soy protein daily. However, this is inconsistent with other meta-analyses and letters were written to the editor of the paper to reanalyse the data due to several reasons 18,19. Soy protein and Soy isoflavones are not found to be estrogenic or androgenic; this appears to be a common finding 20,21. That solves our mystery. What about gynaecomastia? Strangely, one case study demonstrated that a 60-year old man had extremely high oestradiol concentrations and gynaecomastia which resolved with soy milk cessation 22. Thus, there may be inter-individuality with results, or other factors contributed which the clinical case study could not account for. However, this is with soy milk and not soy protein. The literature is less clear with soy milk. Regardless, n = 1 does not garner much statistical power and thus fails to motivate me in thinking much of soy milk. A further critical examination of the clinical evidence once again demonstrates that soy doesn’t have feminising effects on men (though, in some rodents it can) 23. So, what’s the verdict? Well, it’s very likely soybean protein, soy milk, and isoflavone are not going to adversely affect your sex-steroid profile, nor fertility.
What about the tap water; “I hear there’s birth control hormones in it!?”. Well, there has been a recent observation of feminisation of aquatic animals in some countries 24. This has given worries about oestrogenic compounds being present in water supplies, but can they reach drinking water? Well, one study performed an analysis on both children and adults and found that the amount of total oestrogens present in drinking water are not likely to be causing adverse effects among US residents, including sensitive subpopulations 25. In Severn Trent Water, in the UK, no hormones or substances that are oestrogenic were detected in drinking water 26. So, do not worry – tap water is safe!
What about other foods? Well, we know phytoestrogens exist, but as you may have noticed from my last blog, they tend to modulate oestrogen metabolism positively. Hormones in dairy food in the USA is however a potential risk 27, but is not one faced in the UK and EU due to food regulations. In USA dairy produce, physiologic concentrations of steroid hormones exist due to hormone use in cattle. This will inevitably cause endocrine disruption to some extent. Okay – so a risk factor for the USA only? What about pregnant cows that make commercial milk? Well, one study demonstrates that the oestrogens in milk from such cows are absorbed and see a suppression in gonadotropin, followed by a decrease of testosterone 28. This study was conducted in Japan, too. Is this a cause for concern? Potentially, especially in children where it could affect development. However, results may be contradictory in some studies. Furthermore, humans have co-evolved with house cows and are used to drinking milk for at least 2000 years without obvious harm 29. Should there be cause for concern now? Dairy is often enriched with vitamin D and calcium, both important for general health and wellbeing. Further studies are needed in this domain, additionally – this may depend on the way the cow was treated, i.e. was it given hormones in large quantities unnecessarily? Personally, I will continue drinking milk and eating cheese, but whether diary is an EDC is up for debate.
The mechanisms in this case that could lead to plausible HPGA disruptions are embedded in, negatively affecting the HPGA feedback loop. This becomes less of an issue to those on TRT, relative to EDCs found in plastics which can affect things more on the gene level. These are only a few examples of EDCs, and stranger ones yet exist. For example. one cross-sectional study in Chinese male farmers suggested that excess fluoride exposure can decrease serum Testosterone levels 30. Please do not stop brushing your teeth though (your dentist, mouth, partner, and wallet will thank you if you keep brushing!).
I could continue the list of EDCs; they are innumerable and present in our everyday life. Could it explain the epidemic of male hypogonadism and feminisation? Perhaps, but it’s hard to establish. What’s clear is there is a trend that points towards huge increases in sedentary activity and worsening diet; both of which contribute to the epidemic of type 2 diabetes and cardiovascular disease 31, which only serve to worsen body composition further and increase the risk of hypogonadism! This, rather than specific ECDs, are more likely to be the culprits of an increased incidence of hypogonadism within our population. Additionally, there is a slowly increasing acceptance that hypogonadism is a male health issue that needs treatment, thus the number of tests of hypogonadism increase, highlighting more hypogonadal cases. Combine this with an increased population size, meaning a higher risk of statistically finding a hypogonadal person, and already you can see that there’s innumerable factors at play and it’s vastly more complicated than one may think.
Conclusion
So, what is the verdict? You are probably best-off avoiding BPA and sticking to other plastics; recycle them, too. Avoid being around fires where possible, they are carcinogenic due to polycyclic aromatic hydrocarbon exposure, regardless 32. Do not heat up PVCs in you can, but feel free to consume soy or milk produce if you wish to. Also feel free to drink tap water without a need to filter it out of fear of any EDCs. Please brush your teeth with toothpaste, too! There are innumerable EDCs present in our everyday life that it’s hard to find one single EDC that contributes more to HPGA disruption than another, and its likely inevitable that you or I encounter it, for they exist in both man made and natures very own products. Do make sure you get plenty of exercise and eat a healthy, well balanced diet that meets your daily energy needs. That way, you’ll likely avoid most endocrine negative effects of EDCs that you encounter.
References
- Gore AC, Chappell VA, Fenton SE, et al. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr Rev. 2015;36(6):1-150. doi:10.1210/er.2015-1010
- Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, et al. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr Rev. 2009;30(4):293-342. doi:10.1210/er.2009-0002
- Rehman S, Usman Z, Rehman S, et al. Endocrine disrupting chemicals and impact on male reproductive health. Transl Androl Urol. 2018;7(3):490-503. doi:10.21037/tau.2018.05.17
- McCullum-Gomez C. The Endocrine Disruption Exchange (TEDX). J Hunger Environ Nutr. 2016;11(3):439-439. doi:10.1080/19320248.2016.1207405
- Lakind JS, Goodman M, Mattison DR. Bisphenol a and indicators of obesity, glucose metabolism/type 2 diabetes and cardiovascular disease: A systematic review of epidemiologic research. Crit Rev Toxicol. 2014;44(2):121-150. doi:10.3109/10408444.2013.860075
- Leonardi A, Cofini M, Rigante D, et al. The effect of bisphenol a on puberty: A critical review of the medical literature. Int J Environ Res Public Health. 2017;14(9). doi:10.3390/ijerph14091044
- Lee DH. Evidence of the possible harm of endocrine-disrupting chemicals in humans: Ongoing debates and key issues. Endocrinol Metab. 2018;33(1):44-52. doi:10.3803/EnM.2018.33.1.44
- Marcoccia D, Pellegrini M, Fiocchetti M, Lorenzetti S, Marino M. Food components and contaminants as (anti)androgenic molecules. Genes Nutr. 2017;12(1). doi:10.1186/s12263-017-0555-5
- Doyle TJ, Bowman JL, Windell VL, McLean DJ, Kim KH. Transgenerational Effects of Di-(2-ethylhexyl) Phthalate on Testicular Germ Cell Associations and Spermatogonial Stem Cells in Mice1. Biol Reprod. 2013;88(5):112. doi:10.1095/biolreprod.112.106104
- Faure AC, Viel JF, Bailly A, Blagosklonov O, Amiot C, Roux C. Evolution of sperm quality in men living in the vicinity of a municipal solid waste incinerator possibly correlated with decreasing dioxins emission levels. Andrologia. 2014;46(7):744-752. doi:10.1111/and.12143
- Jurewicz J, Hanke W, Radwan M, Bonde J. Environmental factors and semen quality. Int J Occup Med Environ Health. 2009;22(4):305-329. doi:10.2478/v10001-009-0036-1
- Polychlorinated Biphenyl – an overview | ScienceDirect Topics. https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/polychlorinated-biphenyl. Accessed May 21, 2020.
- Akingbemi BT, Ge R, Klinefelter GR, Zirkin BR, Hardy MP. Phthalate-induced Leydig cell hyperplasia is associated with multiple endocrine disturbances. Proc Natl Acad Sci U S A. 2004;101(3):775-780. doi:10.1073/pnas.0305977101
- Wetherill YB, Akingbemi BT, Kanno J, et al. In vitro molecular mechanisms of bisphenol A action. Reprod Toxicol. 2007;24(2):178-198. doi:10.1016/j.reprotox.2007.05.010
- Messinezy M, Pearson TC. Polycythemia. Mol Aspects Med. 1996;17(2):189-207. doi:10.5005/jp/books/12794_35
- Li YR, Li J, Zhao SD, et al. Meta-analysis of shared genetic architecture across ten pediatric autoimmune diseases. Nat Med. 2015;21(9):1018-1027. doi:10.1038/nm.3933
- Lee HJ, Chattopadhyay S, Gong EY, Ahn RS, Lee K. Antiandrogenic effects of bisphenol A and nonylphenol on the function of androgen receptor. Toxicol Sci. 2003;75(1):40-46. doi:10.1093/toxsci/kfg150
- Goodin S, Shen F, Shih WJ, et al. Clinical and biological activity of soy protein powder supplementation in healthy male volunteers. Cancer Epidemiol Biomarkers Prev. 2007;16(4):829-833. doi:10.1158/1055-9965.EPI-06-0882
- Messina M, Hamilton-Reeves J, Kurzer M, Phipps W. Effect of soy protein on testosterone levels [1]. Cancer Epidemiol Biomarkers Prev. 2007;16(12):2795. doi:10.1158/1055-9965.EPI-07-2543
- Haun CT, Mobley CB, Vann CG, et al. Soy protein supplementation is not androgenic or estrogenic in college-aged men when combined with resistance exercise training. Sci Rep. 2018;8(1). doi:10.1038/s41598-018-29591-4
- Hamilton-Reeves JM, Vazquez G, Duval SJ, Phipps WR, Kurzer MS, Messina MJ. Clinical studies show no effects of soy protein or isoflavones on reproductive hormones in men: Results of a meta-analysis. Fertil Steril. 2010;94(3):997-1007. doi:10.1016/j.fertnstert.2009.04.038
- Martinez J, Lewi JE. An unusual case of gynecomastia associated with soy product consumption. Endocr Pract. 2008;14(4):415-418. doi:10.4158/EP.14.4.415
- Messina M. Soybean isoflavone exposure does not have feminizing effects on men: A critical examination of the clinical evidence. Fertil Steril. 2010;93(7):2095-2104. doi:10.1016/j.fertnstert.2010.03.002
- Wise A, O’Brien K, Woodruff T. Are oral contraceptives a significant contributor to the estrogenicity of drinking water? Environ Sci Technol. 2011;45(1):51-60. doi:10.1021/es1014482
- Caldwell DJ, Mastrocco F, Nowak E, et al. An assessment of potential exposure and risk from estrogens in drinking water. Environ Health Perspect. 2010;118(3):338-344. doi:10.1289/ehp.0900654
- Fawell JK, Sheahan D, James HA, Hurst M, Scott S. Oestrogens and oestrogenic activity in raw and treated water in Severn Trent Water. Water Res. 2001;35(5):1240-1244. doi:10.1016/S0043-1354(00)00367-5
- Malekinejad H, Rezabakhsh A. Hormones in Dairy Foods and Their Impact on Public Health. Iran J Public Health. 2015;44(6):742-758. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4524299/. Accessed May 16, 2020.
- Maruyama K, Oshima T, Ohyama K. Exposure to exogenous estrogen through intake of commercial milk produced from pregnant cows. Pediatr Int. 2010;52(1):33-38. doi:10.1111/j.1442-200X.2009.02890.x
- Journal Club. Breast Care. 2010;5(1):44-46. doi:10.1159/000277938
- Duan L, Zhu J, Wang K, et al. Does Fluoride Affect Serum Testosterone and Androgen Binding Protein with Age-Specificity? A Population-Based Cross-Sectional Study in Chinese Male Farmers. Biol Trace Elem Res. 2016;174(2):294-299. doi:10.1007/s12011-016-0726-z
- Panahi S, Tremblay A. Sedentariness and Health: Is Sedentary Behavior More Than Just Physical Inactivity? Front Public Heal. 2018;6. doi:10.3389/fpubh.2018.00258
- Stec AA, Dickens KE, Salden M, et al. Occupational Exposure to Polycyclic Aromatic Hydrocarbons and Elevated Cancer Incidence in Firefighters. Sci Rep. 2018;8(1). doi:10.1038/s41598-018-20616-6